39 secondary labeling requirements
Rules for Proper Secondary Container Labeling - HSI Secondary Container Label Requirements Employers must make sure each container of hazardous chemicals in the workplace is labeled, tagged, or marked with either of the following: All the specific information for the labels on shipped containers. PDF Annex 9 Guidelines on packaging for pharmaceutical products The requirements to be met by pharmaceutical packaging and pack- aging materials as described in compendia (pharmacopoeias) and standards (e.g. those of the International Organization for Standard- ization (ISO)) must be considered only as general in character.
PDF WHAT IS GHS? - Florida International University SECONDARY CONTAINER LABELING: Secondary containers are usually smaller containers, such as spray bottles, jugs, aliquots, or jars that chemicals are transferred to from the primary container once within the workplace. -According to OSHA, secondary containers must comply with the labeling requirements.
Secondary labeling requirements
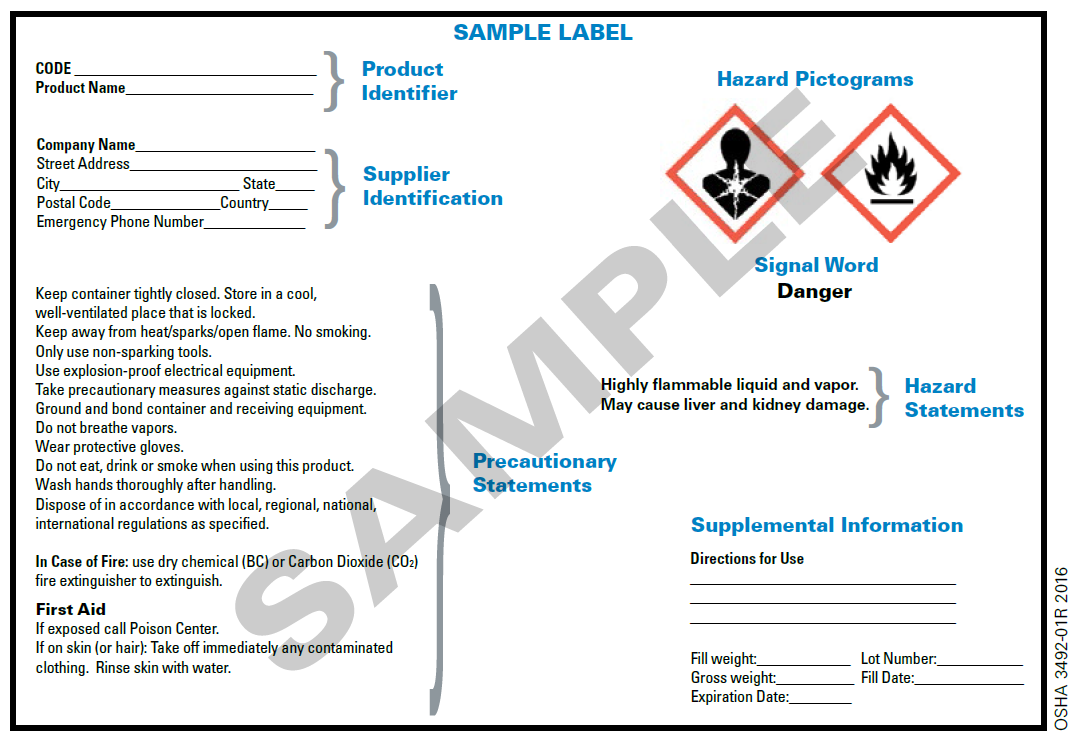
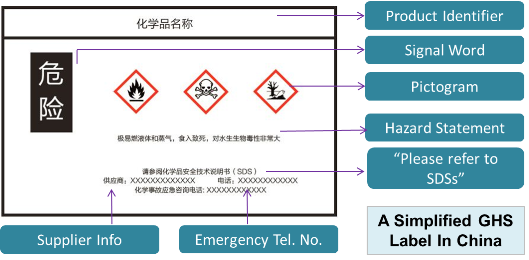
Hazard Communication - Globally Harmonized System | Occupational Safety ... In 2003, the United Nations (UN) adopted the Globally Harmonized System of Classification and Labeling of Chemicals (GHS). The GHS includes criteria for the classification of health, physical and environmental hazards, as well as specifying what information should be included on labels of hazardous chemicals as well as safety data sheets. What Information Is Required On Secondary Container Labels? - XO Safety While chemical shipping containers must have the full GHS label, OSHA provides employers with a lot of flexibility to create their own secondary container labeling systems. OSHA Requirements for Secondary Container Labels OSHA requires secondary container labels to have the full GHS label, or: Packaging and Labeling - Food and Drug Administration Labeling requirements include: ¨ Identifying all tamper-evident features and any capsule sealing technology ¨ Placing labeling prominently on the package ¨ Placing labeling to be unaffected...
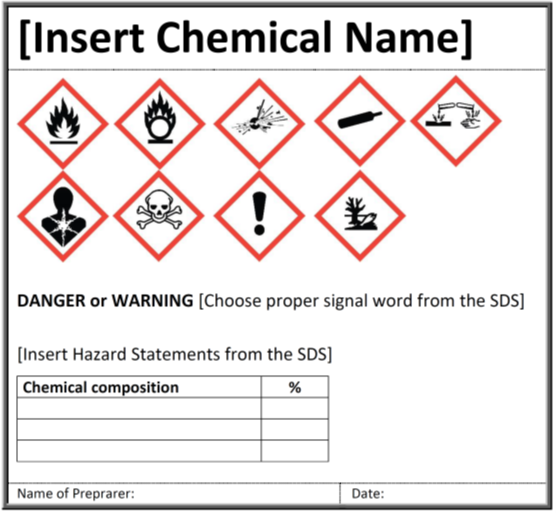
Secondary labeling requirements. eCFR :: 21 CFR Part 211 Subpart G -- Packaging and Labeling Control Labeling and packaging materials shall be representatively sampled, and examined or tested upon receipt and before use in packaging or labeling of a drug product. ( b) Any labeling or packaging materials meeting appropriate written specifications may be approved and released for use. Hazard communication - Oregon Occupational Safety and Health Rules with requirements We get many calls and emails from employers asking the same question. It begins: "Which of your rules have requirements for…" You can finish the sentence with words like "recordkeeping," "employee training," and "written documents." Labeling small containers - Occupational Safety and Health Administration Response: 29 CFR 1910.1200 (f) (1) requires the chemical manufacturer, importer, or distributor to ensure that each container of hazardous chemicals leaving the workplace is labeled, tagged or marked with the following information: (1) a product identifier; (2) signal word; (3) hazard statement (s); (4) pictogram (s); (5) precautionary statement … Labeling secondary containers - JJKellerSafety.com What information is required on secondary container labels? If the chemical is going to be used only "in house," then the container is to be labeled in accordance with 1910.1200 (f) (6) . Label it with the product identifier, words, pictures, symbols, or a combination thereof. You can use HMIS or NFPA labels, or your own system, for in-house ...
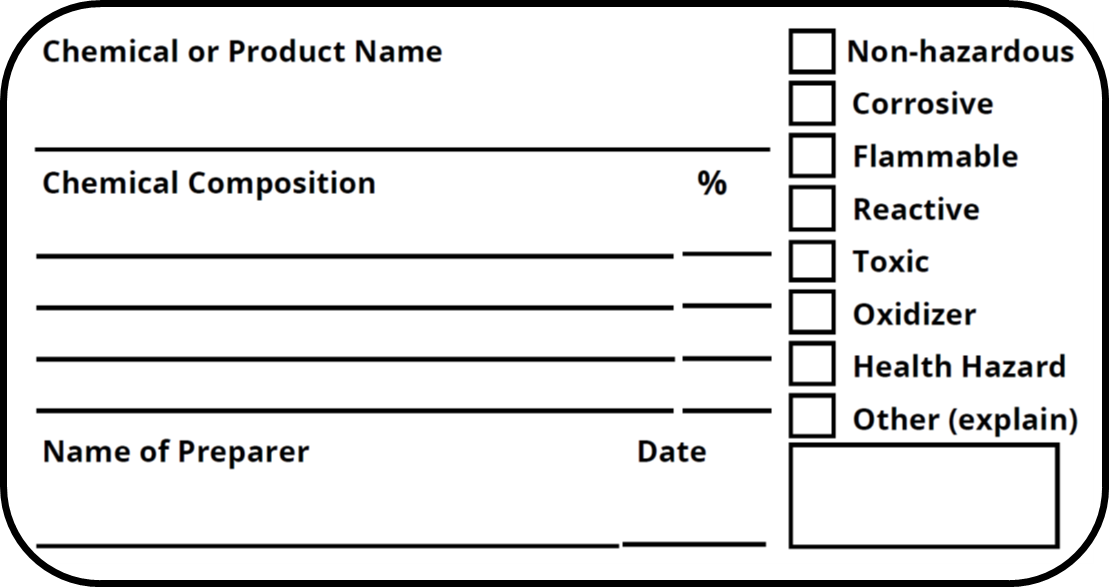
Secondary Containers and Service Containers for Pesticides EPA Recommendations for Labeling Secondary and Service Containers Although the Agency does not require labels on secondary and service containers, the Department of Transportation (DOT) and Occupational Safety and Health Administration (OSHA) requirements may apply. California Code of Regulations, Title 8, Section 5194. Hazard ... 1. an employer employing fewer than ten employees; 2. any city, county, or district or any department or agency thereof or the state or any department or agency thereof or the federal government or any department or agency thereof; 3. any entity in its operation of a public water system as defined in Health and Safety Code Section 4010.1. What Is a Secondary Label and What Makes It OSHA Compliant? Steps to Take to Make Sure the Secondary Label is OSHA Compliant OSHA requires that you label hazardous chemicals using six indicators. These indicators communicate the physical, health, and environmental hazards associated with a particular chemical. Some of the steps that will increase your compliance with these guidelines include: Does OSHA Require Secondary Containers of Water to be Labeled? Aug 19, 2015. When chemicals or hazardous materials are transferred from a parent container to a secondary container, OSHA requires the secondary container to be labeled. This secondary container label should contain all of the safety information from the parent container, so that workplace hazards are clearly communicated to everyone.
Secondary Containers | Environmental Health, Safety and Emergency ... Except for a few cases, secondary containers must be labeled. IF IN DOUBT, LABEL IT! One common case where you do not have to label a secondary container is if the container is portable and will be used immediately by the person who transferred the chemical into that container. For example, if you pour a concentrated disinfectant into a bucket ... GHS Label Requirements: What Information to Include - MPC GHS-compliant labels contain six main elements.Note that these requirements apply to primary containers (which includes the containers received from the manufacturer), but not specifically to secondary containers (such as smaller jars or spray bottles that hold chemicals transferred from the primary container). FDA's Labeling Resources for Human Prescription Drugs | FDA DailyMed: NIH's labeling tool designed to search over 140,000 labeling documents for prescription drugs, nonprescription drugs, animal drugs, and other products (e.g., animal nonprescription and ... Hazard Communication - Overview | Occupational Safety and Health ... Labels: Chemical manufacturers and importers will be required to provide a label that includes a harmonized signal word, pictogram, and hazard statement for each hazard class and category. Precautionary statements must also be provided. Safety Data Sheets: Will now have a specified 16-section format.
Clinical Labeling of Medicinal Products: EU Clinical Trial ... - ISPE Clinical labeling requirements for the "period of use" do not only apply to the outer (secondary) packaging but also apply to the immediate (primary) packaging.. This aspect could have a high level impact: this way we can foresee that periodic updates of the "period of use", commonly applied in the course of a clinical study when new ...

GHS Hazard Secondary Container Label Stickers 3 X 4 Inches - SDS/OSHA Stickers for Chemical Safty Data MSDS/HMIG Write-in Labels 250/Roll
OSHA Requirements for Labeling Secondary Containers Secondary label regulations only apply to containers used inside the facility. If you're repackaging bulk products for sale, these new containers need to meet all OSHA requirements for primary containers. Creating Labels that Meet Both United States and Canadian Labeling Requirements
Secondary Container Labels 101: HazCom and WHMIS - ERA Environmental In the US secondary container labels are required when operations in a work-place setting includes the transferring of smaller amounts from the original container to a secondary container such as a beaker, flask, or bottle. These secondary labels need to comply with OSHA's HCS.
Compliance FAQs: Packaging and Labeling in the US | NIST Labeling requirements related to legal metrology (i.e., products and commodities sold in package form by weight, measure or count) must comply with The Fair Packaging and Labeling Act (FPLA) and Uniform Packaging and Labeling Regulation (UPLR), NIST Handbook 130-Current Edition ).
Chemical Container Labels | EHS - University of Washington EH&S has designed four secondary chemical container label templates for your use. Templates A and B are PDF fillable forms. Templates C and D are Word documents that show GHS pictograms. Templates A, B and C are formatted for printing on Avery 5163 (2" x 4" label, 8 labels, 8½" x 11" page). All templates can be resized to fit smaller ...
4 Quick Tips to Help You Ace OSHA Secondary Container Labeling OSHA labeling requirements for secondary containers (that do not qualify for exemption) are outlined in HazCom standard 1910.1200 (f) (6) (ii) and summarized listed below. Product Name/Identifier. This should match the product identifier on the safety data sheet. General Hazard Statement.
Quality System Regulation Labeling Requirements | FDA Various sections of the QS regulation have an impact on labeling: Section 21 CFR 820.80 (b) requires the inspection and testing of incoming materials including labeling; and 21 CFR 820.70 (f ...
GHS Labeling Requirements: The Definitive Guide [2021 Update ... - Luminer These GHS label requirements are meant to succinctly communicate key information about what is inside. However, the GHS takes into consideration that, sometimes, it's not possible to keep workers safe with only these six label requirements, which is why it also allows for supplemental information. 1. Product Identifier
PDF Guidelines for Labeling Secondary Containers - UGA Non-Hazardous Substance Labeling Secondary containers for non-hazardous substances (e.g., saline, feed, water) must be labeled with the name of the substance, and dated if perishable or sterile. Perishable items must be labeled with an expiration date*. When a container is dated, the date must be clearly distinguished as either a fill date,
Download secondary chemical container labels | EHS Secondary Container Label 1 Secondary Container Label 2 Secondary Container Label 3 Exemption: Secondary labels are not required when the container is under the direct control of the person who transferred or prepared the solution and all of it will be consumed during the same work shift.
PDF BRIEF - Occupational Safety and Health Administration labeling requirements as a part of its recent revision of the Hazard Communication Standard, 29 CFR 1910.1200 (HCS), bringing it into alignment with the United Nations' Globally Harmonized System of Classification and Labelling of Chemicals (GHS). These changes will help ensure improved quality and consistency in the classification and labeling
Labeling of Secondary Containers | Occupational Safety and Health ... Labeling of Secondary Containers Standard Number: 1910.1200 (f) (6) (ii) OSHA requirements are set by statute, standards and regulations. Our interpretation letters explain these requirements and how they apply to particular circumstances, but they cannot create additional employer obligations.
Packaging and Labeling - Food and Drug Administration Labeling requirements include: ¨ Identifying all tamper-evident features and any capsule sealing technology ¨ Placing labeling prominently on the package ¨ Placing labeling to be unaffected...
What Information Is Required On Secondary Container Labels? - XO Safety While chemical shipping containers must have the full GHS label, OSHA provides employers with a lot of flexibility to create their own secondary container labeling systems. OSHA Requirements for Secondary Container Labels OSHA requires secondary container labels to have the full GHS label, or:
Hazard Communication - Globally Harmonized System | Occupational Safety ... In 2003, the United Nations (UN) adopted the Globally Harmonized System of Classification and Labeling of Chemicals (GHS). The GHS includes criteria for the classification of health, physical and environmental hazards, as well as specifying what information should be included on labels of hazardous chemicals as well as safety data sheets.





























Post a Comment for "39 secondary labeling requirements"